 
Interestingly, a strong correlation between O 2 and H 2O has been identified, indicating the O 2 formation is linked to H 2O in the comet. Recently, abundant molecular O 2 in the coma of comet 67P/Churyumov–Gerasimenko, which is dominated by H 2O, CO, and CO 2, has been detected 13. In contrast, photodissociation of H 2O, one of the dominant oxygen carriers 12, has long been assumed to proceed mainly to produce hydroxyl (OH) and hydrogen (H) atom primary products, and contribute limitedly to the O 2 production 9. These findings provide new insights into the sources of O 2 in Earth’s early atmosphere. Direct O 2 production pathways via VUV photodissociation of CO 2 10 and dissociative electron attachment to CO 2 11 have recently been identified. Once this happens, the salt is dissolved, resulting in a homogeneous solution.Where M is a third body to carry off the excess energy in the reaction process. After the salt compounds are pulled apart, the sodium and chloride atoms are surrounded by water molecules, as this diagram shows. Water molecules pull the sodium and chloride ions apart, breaking the ionic bond that held them together. Essentially, a tug-of-war ensues with the water molecules winning the match. The positively-charged side of the water molecules are attracted to the negatively-charged chloride ions and the negatively-charged side of the water molecules are attracted to the positively-charged sodium ions. When salt is mixed with water, the salt dissolves because the covalent bonds of water are stronger than the ionic bonds in the salt molecules. Likewise, a water molecule is ionic in nature, but the bond is called covalent, with two hydrogen atoms both situating themselves with their positive charge on one side of the oxygen atom, which has a negative charge. The bonds in salt compounds are called ionic because they both have an electrical charge-the chloride ion is negatively charged and the sodium ion is positively charged. It also depicts how a charge, such as on an ion (Na or Cl, for example) can interact with a water molecule.Ĭredit: Mariana Ruiz Villarreal, CK-12 Foundation Why salt dissolves in waterĪt the molecular level, salt dissolves in water due to electrical charges and due to the fact that both water and salt compounds are polar, with positive and negative charges on opposite sides in the molecule. This diagram shows the positive and negative parts of a water molecule. That is where water helps out being such a great solvent, water washing through the kidneys dissolves these substances and sends them on the way out of our bodies. But, the kidneys have got to get rid of these substances after they accumulate them. The kidneys are responsible for filtering out substances that enter our bodies from the foods and drinks we consume. Our own kidneys and water's solvent properties make a great pair in keeping us alive and healthy. 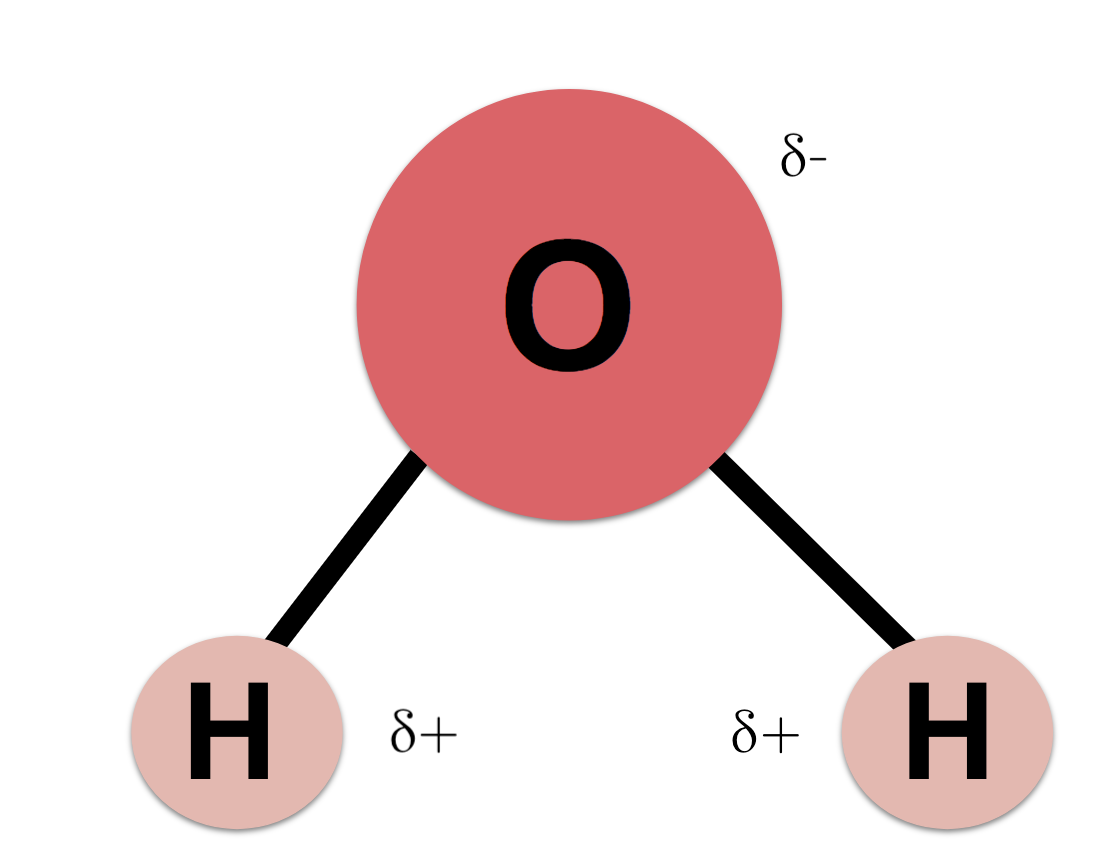
Water can become so heavily attracted to a different compound, like salt (NaCl), that it can disrupt the attractive forces that hold the sodium and chloride in the salt compound together and, thus, dissolve it. This allows the water molecule to become attracted to many other different types of molecules. Water molecules have a polar arrangement of oxygen and hydrogen atoms-one side (hydrogen) has a positive electrical charge and the other side (oxygen) had a negative charge. It is water's chemical composition and physical attributes that make it such an excellent solvent. It means that wherever water goes, either through the air, the ground, or through our bodies, it takes along valuable chemicals, minerals, and nutrients. This is important to every living thing on earth. Water is called the "universal solvent" because it is capable of dissolving more substances than any other liquid. Visit Media to see details.ĭid you know you can dissolve the M off of an M&M?All you have to do it put a few M&Ms in water with the M side up and observe what happens! Sources/Usage: Some content may have restrictions. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
